How Does a Pacemaker Work?
 Pacemakers
Pacemakers
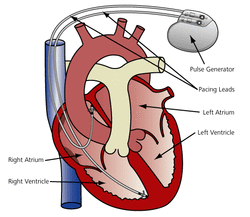
A pacemaker is a small device that is implanted inside the chest area to aid the heart in maintaining a proper heart rate. It sends small electric pulses to the heart to keep blood pumping and regulate a correct heartbeat. Some devices require an open-heart surgery to take place while newer devices are small enough to where they can be inserted through an incision in the leg. Another job of this device is to help alleviate issues with arrhythmia such as fatigue and fainting.
History
The first instance where a pacemaker was used was in 1899. The device was created by J.A. McWilliam. During a period of irregular heart rates noted by patients, this device was used externally to return a systolic heart rate of 60 to 70 beats per minute. While this is still slower than a normal functioning heart, it is sufficient enough to sustain life.
Developments were made by 1926 to improve the first pacemaker. By this time the device was made portable and able to create a sustained heartbeat of 80 to 120 beats per minute which is more in the normal range. With this device, one “pole” would touch a lighting point while the other would touch a skin pad that was soaked in a salt solution.
Fast forward a few decades to 1958 and you have devices that could be implanted inside the body. These meet medical device compliance in the sense that they must be made to not short, cause electrocution or send a charge that is too strong for the patient’s heart. Further developments have made the device so small that it is now the size of a multivitamin.
What a Pacemaker is made from
A pacemaker is constructed from electrodes that meet pharmaceutical compliance regulations and several other components. The electrodes deliver the pulses to force the heart to contract. The other materials used include polyurethane, titanium, titanium alloys, lithium/ion batteries, and sterilizable leads. A motherboard is also required to send the frequency for the pulses.
How it Works
Each pacemaker is programmed to treat a patient specifically. While some are the sole support of a heart in reference to creating pulses or beats, others are an auxiliary or backup device that can sense when to send a pulse. The leads touch the heart and send the “shock” to the heart from the motherboard or control center inside the module.
Compliance Standards in Canada
Medical device compliance in Canada is responsible for classifying medical devices and pharmaceuticals into categories. Devices in Class 1 require no specific license since they have little or no risk. Strict security regulations and inspection processes along with material compositions are in place when it comes to Class 3 and Class 4 devices.
Compliance Standards in the United Kingdom
The medical device compliance in the UK follows the same regulations that are generally stated for Europe. This includes similar classifications to Canada with a broader range of devices, clarifications, packaging policies and inspection processes. Essentially, the longer a device is slated to be in the body, the higher classification it receives. Bodily contact with medical devices is highly regulated in European countries.
Classification Standards:
· Requirement of an external energy source
· Viewable CE mark on the packaging
· Effect on the central circulation and nervous systems
· Combination with other medicinal products
· Overall diagnostic impact to the body
· Packaging guidelines are followed
· Product is sterile upon delivery
Someone of a notable stature must sign a certificate of conformity for a manufacturer of medical devices to be a distributor of medical devices in the UK.
Compliance Standards in the United States
In regards to medical device compliance in the US, three main categories exist. Each category is outlined in detail. Category 1 or general controls includes class 1 and class 2 devices. These controls regulate items such as device registration, ensuring that life sustaining items are not in class 1 or 2, and do not pose a significant risk to human life. Moving up to category 2, these devices are those that include a special control that has to be programmed, monitored or regulated from time-to-time. Category 3 refers to general devices with pre-market approvals to be used on a preliminary or regulated-study basis. Each of these categories is heavily monitored for FDA and federal standards.
The pacemaker is a lifesaving medical device that has been in use for well over a century all around the world. Advancements have come very far since the first device was created in 1899. The surgical process has become far less invasive and requires less recovery time. Intracardial devices were introduced in 2013 and have proven to be very successful and require far less maintenance. The batteries used in new-age devices can now outlive patients. Fewer device malfunctions occur and fewer patients require an adjustment of their device to sustain a normal heartbeat.
History
The first instance where a pacemaker was used was in 1899. The device was created by J.A. McWilliam. During a period of irregular heart rates noted by patients, this device was used externally to return a systolic heart rate of 60 to 70 beats per minute. While this is still slower than a normal functioning heart, it is sufficient enough to sustain life.
Developments were made by 1926 to improve the first pacemaker. By this time the device was made portable and able to create a sustained heartbeat of 80 to 120 beats per minute which is more in the normal range. With this device, one “pole” would touch a lighting point while the other would touch a skin pad that was soaked in a salt solution.
Fast forward a few decades to 1958 and you have devices that could be implanted inside the body. These meet medical device compliance in the sense that they must be made to not short, cause electrocution or send a charge that is too strong for the patient’s heart. Further developments have made the device so small that it is now the size of a multivitamin.
What a Pacemaker is made from
A pacemaker is constructed from electrodes that meet pharmaceutical compliance regulations and several other components. The electrodes deliver the pulses to force the heart to contract. The other materials used include polyurethane, titanium, titanium alloys, lithium/ion batteries, and sterilizable leads. A motherboard is also required to send the frequency for the pulses.
How it Works
Each pacemaker is programmed to treat a patient specifically. While some are the sole support of a heart in reference to creating pulses or beats, others are an auxiliary or backup device that can sense when to send a pulse. The leads touch the heart and send the “shock” to the heart from the motherboard or control center inside the module.
Compliance Standards in Canada
Medical device compliance in Canada is responsible for classifying medical devices and pharmaceuticals into categories. Devices in Class 1 require no specific license since they have little or no risk. Strict security regulations and inspection processes along with material compositions are in place when it comes to Class 3 and Class 4 devices.
Compliance Standards in the United Kingdom
The medical device compliance in the UK follows the same regulations that are generally stated for Europe. This includes similar classifications to Canada with a broader range of devices, clarifications, packaging policies and inspection processes. Essentially, the longer a device is slated to be in the body, the higher classification it receives. Bodily contact with medical devices is highly regulated in European countries.
Classification Standards:
· Requirement of an external energy source
· Viewable CE mark on the packaging
· Effect on the central circulation and nervous systems
· Combination with other medicinal products
· Overall diagnostic impact to the body
· Packaging guidelines are followed
· Product is sterile upon delivery
Someone of a notable stature must sign a certificate of conformity for a manufacturer of medical devices to be a distributor of medical devices in the UK.
Compliance Standards in the United States
In regards to medical device compliance in the US, three main categories exist. Each category is outlined in detail. Category 1 or general controls includes class 1 and class 2 devices. These controls regulate items such as device registration, ensuring that life sustaining items are not in class 1 or 2, and do not pose a significant risk to human life. Moving up to category 2, these devices are those that include a special control that has to be programmed, monitored or regulated from time-to-time. Category 3 refers to general devices with pre-market approvals to be used on a preliminary or regulated-study basis. Each of these categories is heavily monitored for FDA and federal standards.
The pacemaker is a lifesaving medical device that has been in use for well over a century all around the world. Advancements have come very far since the first device was created in 1899. The surgical process has become far less invasive and requires less recovery time. Intracardial devices were introduced in 2013 and have proven to be very successful and require far less maintenance. The batteries used in new-age devices can now outlive patients. Fewer device malfunctions occur and fewer patients require an adjustment of their device to sustain a normal heartbeat.
