How Does an Artificial Heart Valve Work?
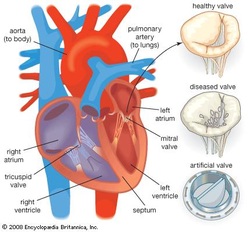 Artificial Valves for the Heart
Artificial Valves for the Heart
Artificial heart valves are used for a variety of reasons in heart patients. The most common is to replace a clogged valve or one that is no longer functioning. Consider the heart as being the engine of a car, the valves are needed to open and close the aorta and ventricles of the heart. Open heart surgery is required to alleviate this issue. In most cases, only one valve needs to be replaced. Medical compliance standards must be met by all artificial devices that are to be implanted inside the body, especially those dealing with the heart.
History
Beginning development in the 1940s, the first artificial or mechanical heart valve was implanted in 1952. The first patient was a 30-year old female and the device implanted into her was a ball-type valve. This valve was designed by Dr. Charles Hufnagel. While this valve did help the patient it had some disadvantages since it was only able to be implanted into the descending aorta. It would not work for the heart cavity area itself.
The 1960s led to improvements in design and implantation process. A valve produced by Star-Edwards was developed in the same general idea, a ball valve, which modified Hufnagel’s original design. The variations for this design were a caged exterior and slightly smaller in size. These changes made the ball valve able to be implanted into the heart rather than on the descending portion.
Continuing with developments and improvements, in the late 1960s, tilting discs were developed for the valve. What this did for the ball valve design was allow blood to flow more freely. It was also a way to reduce damaging red blood cells in the filtration process. Although initially approved for medical use, these valves proved to be weak when it came to longevity of use. After a few years of continual use, the valves gave out, requiring a patient to go through another surgical procedure.
In 1979, Bileaflet valves were introduced to surgeons. The inlet strut on this device was made of a stronger material in order to make the tilting disc stronger and work for a longer period of time. Many modern heart valves use this same design. There is one drawback here, and that is a minor amount of backflow, which has not proved to be a big problem.
Types
There are several types of artificial heart valves in use for medical implantation reasons. Manufactured heart valves are an option but so are donor heart valves and tissue valves. Depending on the needs of the patient, and availability of donor and tissue options, those may be used as a more naturally functioning heart valve. These do require some monitoring after the procedure to ensure that the donor valve is working properly and to ensure that the tissue is remaining attached.
Manufactured valves must meet medical compliance in the US by not including mesh. These valves must also include surgically approved metal and plastic or ceramic products to ensure that no reaction to the materials occurs. These must also be non-electrical conducting materials. The same protocols are in place for medical compliance in the UK and medical compliance in Canada. Additional requirements include the requirement of blood types, DNA markers and good physical health being available for the right donor. The tissue used must be viable, healthy and durable enough to sustain the lifespan of the patient.
What the Valves do
There are four types of valves; aortic, pulmonary, tricuspid and mitral valves. Artificial valves are designed to increase the flow of blood to and from the heart. These are inserted into the heart valve or section of the heart that needs the blood flow increased. What happens when a valve is inserted is that it is tested and viewed for functionality while monitoring the heart rate and statistics of the patient.
The pharmaceutical compliance standards are very strict for heart valves. This is due to the function that they perform and the period of time that the device has to last. In younger patients, most surgeons opt to use a manufactured valve since these last the longest. Biological and donor valves will last for several years but are often a temporary solution while heart physicians explore other underlying issues in a patient.
History
Beginning development in the 1940s, the first artificial or mechanical heart valve was implanted in 1952. The first patient was a 30-year old female and the device implanted into her was a ball-type valve. This valve was designed by Dr. Charles Hufnagel. While this valve did help the patient it had some disadvantages since it was only able to be implanted into the descending aorta. It would not work for the heart cavity area itself.
The 1960s led to improvements in design and implantation process. A valve produced by Star-Edwards was developed in the same general idea, a ball valve, which modified Hufnagel’s original design. The variations for this design were a caged exterior and slightly smaller in size. These changes made the ball valve able to be implanted into the heart rather than on the descending portion.
Continuing with developments and improvements, in the late 1960s, tilting discs were developed for the valve. What this did for the ball valve design was allow blood to flow more freely. It was also a way to reduce damaging red blood cells in the filtration process. Although initially approved for medical use, these valves proved to be weak when it came to longevity of use. After a few years of continual use, the valves gave out, requiring a patient to go through another surgical procedure.
In 1979, Bileaflet valves were introduced to surgeons. The inlet strut on this device was made of a stronger material in order to make the tilting disc stronger and work for a longer period of time. Many modern heart valves use this same design. There is one drawback here, and that is a minor amount of backflow, which has not proved to be a big problem.
Types
There are several types of artificial heart valves in use for medical implantation reasons. Manufactured heart valves are an option but so are donor heart valves and tissue valves. Depending on the needs of the patient, and availability of donor and tissue options, those may be used as a more naturally functioning heart valve. These do require some monitoring after the procedure to ensure that the donor valve is working properly and to ensure that the tissue is remaining attached.
Manufactured valves must meet medical compliance in the US by not including mesh. These valves must also include surgically approved metal and plastic or ceramic products to ensure that no reaction to the materials occurs. These must also be non-electrical conducting materials. The same protocols are in place for medical compliance in the UK and medical compliance in Canada. Additional requirements include the requirement of blood types, DNA markers and good physical health being available for the right donor. The tissue used must be viable, healthy and durable enough to sustain the lifespan of the patient.
What the Valves do
There are four types of valves; aortic, pulmonary, tricuspid and mitral valves. Artificial valves are designed to increase the flow of blood to and from the heart. These are inserted into the heart valve or section of the heart that needs the blood flow increased. What happens when a valve is inserted is that it is tested and viewed for functionality while monitoring the heart rate and statistics of the patient.
The pharmaceutical compliance standards are very strict for heart valves. This is due to the function that they perform and the period of time that the device has to last. In younger patients, most surgeons opt to use a manufactured valve since these last the longest. Biological and donor valves will last for several years but are often a temporary solution while heart physicians explore other underlying issues in a patient.
